There is currently a series of patent disputes surrounding a new method of gene editing called CRISPR-Cas9 (CRISPR), which has huge potential for advancing drug discovery and the treatment of diseases. The system has generated much excitement in the scientific community because it is faster, cheaper, more accurate and more efficient than other existing methods.
Here we explain the current patent situation in the two key geographies (Europe and the US), explore potential future scenarios, and set out what it means for pharma companies seeking to leverage this technology for their R&D programmes.[1]
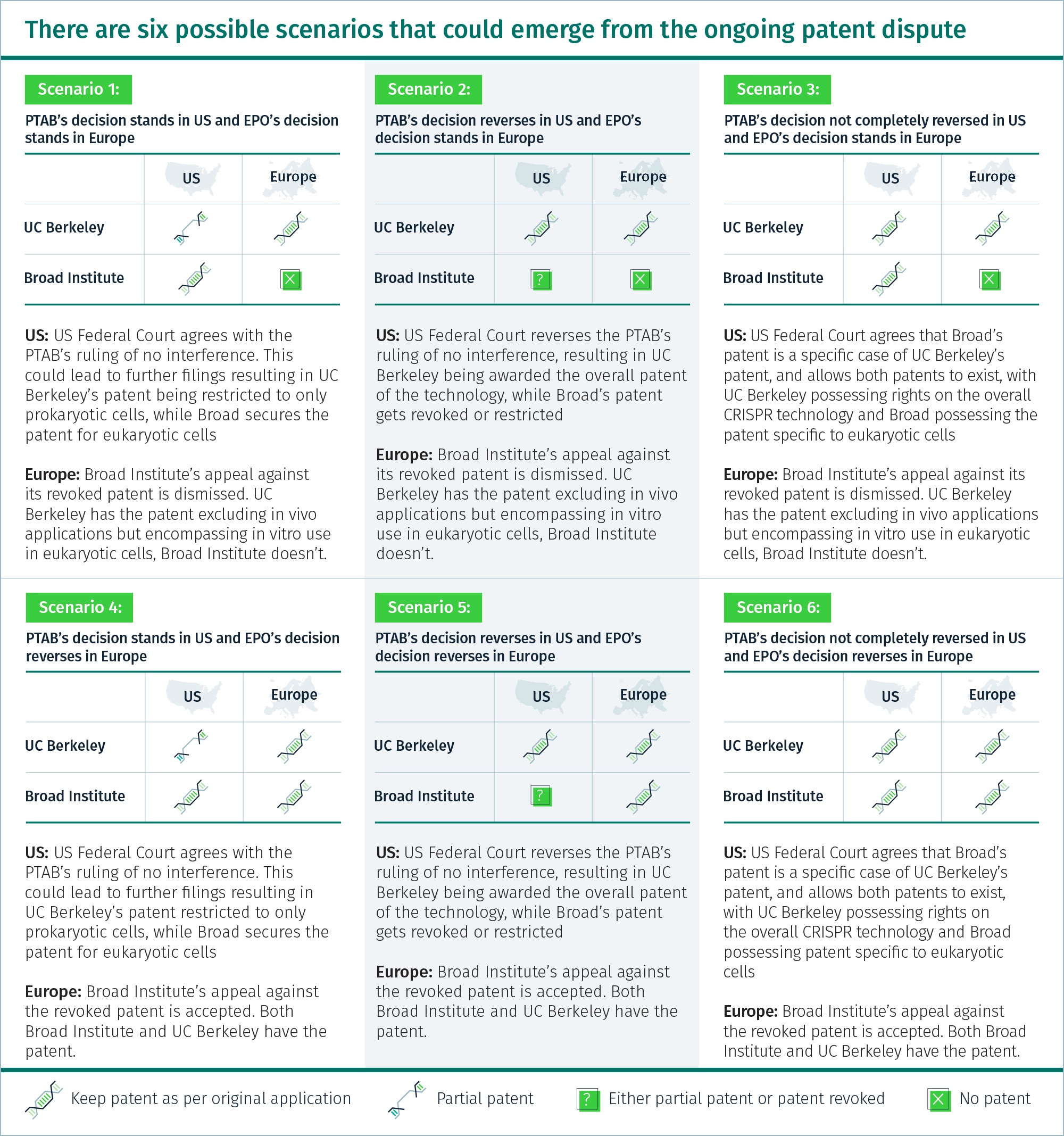
The dispute
Broad Institute and the University of California Berkeley (UC Berkeley) are the two main entities, among a number of others, embroiled in dispute over the patent of the CRISPR technology. The patent row is at different stages in Europe and the US. It is further complicated as entities might be awarded overlapping patents.
In Europe, the patent has been awarded to UC Berkeley for using CRISPR technology in all cell types. Broad Institute’s 10 fundamental CRISPR patents were revoked by the European Patent Office (EPO) in January 2018. The institute made its intentions clear about appealing against the decision but has yet to do it.
In the US, Broad Institute has been granted the patent for CRISPR technology for eukaryotic cells. UC Berkeley had filed for a patent encompassing all organisms before Broad; it has recently been awarded two CRISPR patents with a considerably narrower scope than Broad’s. UC Berkeley filed for interference proceedings with the Patent Trial and Appeal Board (PTAB) and lost. Following this, it approached the US appeals court to reverse PTAB’s decision and is currently awaiting judgment.
Challenges for pharma companies
Different entities holding different patents in different geographies have complicated the licensing procedure. Global pharma companies that are looking at CRISPR technology to bolster their R&D efforts do not have clarity on how to go about licensing it. They cannot use this technology in the commercial space unless they have obtained the requisite licenses.
What does it mean for pharma companies?
Licensing a CRISPR patent could be worth millions of dollars. A mistake in the selection of the entity to partner with and pharmaceutical companies could be looking at massive losses. If either among scenario 2, 3, 5, or 6 materialise, companies which already have a license from an entity could lose money. For example, a pharma company which has a license for commercial use from Broad Institute: in a scenario where Broad’s patent is restricted and UC Berkeley is awarded an overall patent (Scenario 2), the company will have to take another license from UC Berkeley because Broad’s license might not be valid.
A quick scan of the six scenarios and one thing is certain – either Broad Institute or UC Berkeley will have a patent in at least one of the two geographies. This will require obtaining a license from different entities in different geographies, which would eventually mean additional cost. In case of Scenario 3, where both UC Berkeley and Broad Institute have a patent in the US, pharma players will have to obtain a bundled license from both of them for a CRISPR application. For instance, Novartis has partnered with commercial affiliates of UC Berkeley and has invested millions of dollars in getting exclusive license to use the tech in CAR-T cells. Juno Therapeutics, a biopharma company, has bought the same exclusive license from Broad’s commercial affiliate. But if Broad’s patent is upheld in the US, then Novartis would need a license from Juno, and vice versa, to be able to actually use the technology – so, each exclusive licensee would have to grant a sublicense to the other party.
What does the future hold?
The R&D pipeline of pharma players today is dry compared to previous years. CRISPR is a revolutionary technology which can be a very useful R&D tool for these companies, therefore, a head-start in using it could prove hugely beneficial for drug discovery. However, partnering with the wrong entity could lead to significant losses.
Looking at the current patent scenario in the US and Europe, pharma companies that aim to invest in CRISPR discovery will probably need rights from both UC Berkeley and Broad. Initially, some of the big pharmas were betting on just one of the parties, but it is becoming clearer that they might both end up with the necessary rights. And this uncertainty is here to stay for a few more years. Pharma players need to assess whether to wait till the clouds over this patent dispute are completely cleared or if the risk is worth taking to get a head-start in creating a super drug.
[1] Overall there are many different CRISPR patents in existence, and ultimately many institutions will hold many patents in different countries. This blog addresses the dispute for patent to commercially use CRISPR on eukaryotic cells.
To find out how The Smart Cube helps Life Sciences businesses stay ahead, please read about our intelligence and analytics solutions or get in touch. We can help you understand and anticipate the forces and changes influencing your critical procurement categories today and beyond.



